Noble Gas Configuration For Chlorine
4.i: The Art of Deduction- Stable Electron Configurations
- Page ID
- 351223
- Define the octet rule.
The octet rule is a chemical rule of thumb that reflects observation that elements tend to bond in such a way that each atom has viii electrons in its valence beat, giving it the same electronic configuration as a noble gas.
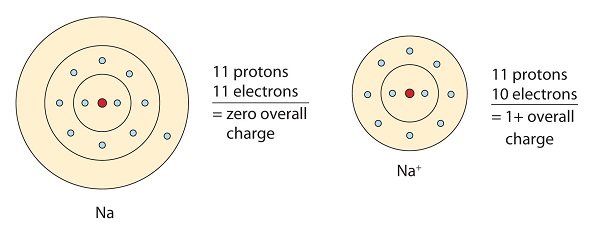
Referring to the octet rule, atoms attempt to get a element of group 0 electron configuration, which is 8 valence electrons. Metals take few electrons in their outer-most orbitals. By losing those electrons, these metals can achieve element of group 0 configuration and satisfy the octet rule. Sodium has one valence electron, so giving it up would outcome in the same electron configuration every bit neon. Due to the loss of electron, a positively charged cation (Na+) called sodium ion is formed.
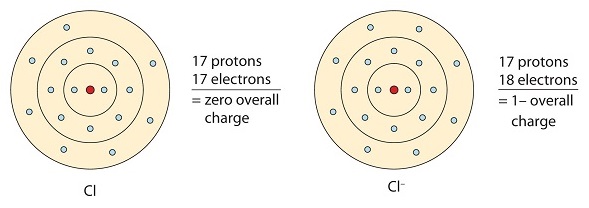
Similarly, nonmetals that have shut to viii electrons in their valence shells tend to readily accept electrons to achieve element of group 0 configuration. Chlorine has seven valence electrons, and so if it takes one information technology will have 8 (an octet). Chlorine has the electron configuration of argon when it gains an electron. The resulting ion (Cl-) is called chloride ion.
The octet dominion could have been satisfied if chlorine gave up all 7 of its valence electrons and sodium took them. In that example, both would have the electron configurations of noble gasses, with a full valence trounce. However, their charges would be much higher. It would be Na7 - and Cl7 +, which is much less stable than Na+ and Cl-. Atoms are more than stable when they have no charge, or a small accuse.
Summary
The octet rule refers to the tendency of atoms to prefer to have 8 electrons in the valence beat out.
Contributor
Organic Chemical science Supplemental_Modules_(Organic_Chemistry)/Fundamentals/Ionic_and_Covalent_Bonds
Noble Gas Configuration For Chlorine,
Source: https://chem.libretexts.org/Courses/Sacramento_City_College/SCC%3A_CHEM_330_-_Adventures_in_Chemistry_(Alviar-Agnew)/04%3A_Chemical_Bonds/4.01%3A_The_Art_of_Deduction-_Stable_Electron_Configurations
Posted by: wyckoffsupuldn.blogspot.com


0 Response to "Noble Gas Configuration For Chlorine"
Post a Comment